The PHQ-2 and the PHQ-8 as Screening Tools for Depression Severity in a Clinical Trial
Category: Research Poster
Author(s): Jayden Mather
Presenter(s): Jayden Mather
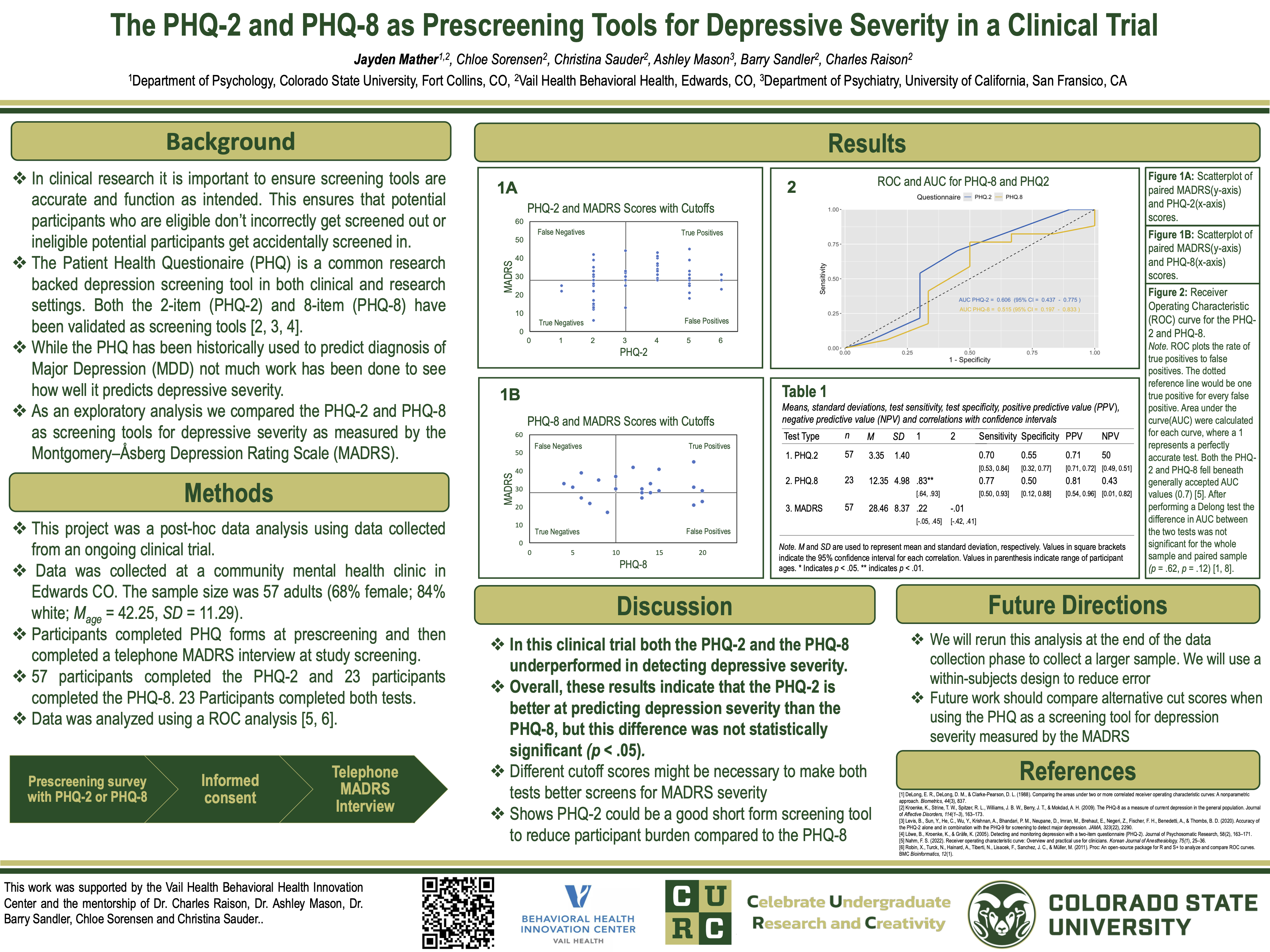
When recruiting subjects for clinical trials, using brief and effective screening tools is important in order to reduce both subject and study team burden. Clinical trials for Major Depressive Disorder (MDD) often use various forms of the Patient Health Questionnaire (PHQ) as a brief screening measure of depressive severity. In an ongoing clinical trial for depression at the Vail Health Behavioral Health Innovation Center, we conducted an exploratory analysis comparing the PHQ-2 and PHQ-8 as screening tools for MDD severity. Potential participants (n = 57) were prescreened with the PHQ-2 (n = 57) and PHQ-8 (n = 23) for depression symptoms. At screening, depressive severity was measured with the clinician-rated Montgomery–Åsberg Depression Rating Scale (MADRS), where a score of 28 or greater was used as a cutoff score. PHQ-2 sensitivity and specificity were 0.70(95% CI [0.53, 0.84]) and 0.55(95% CI [0.32, 0.77]), respectively. PHQ-2 scores had a medium positive correlation with MADRS scores (r = .22, p = 0.12, 95% CI [-.05, .45]). The PHQ-8 sensitivity and specificity were 0.77(95% CI [0.50, 0.93]) and 0.50(95% CI [0.12, 0.88]), respectively. PHQ-8 scores had a very small negative correlation with MADRS scores (r = -.01, p = .97, CI [-.42, .41]). The PHQ-8 had higher sensitivity for detecting the depressive severity, while the PHQ-2 had higher specificity for detecting depression severity.
