Optimization of Bacteriophage Production for Use in Phage Therapy
Category: Research Poster
Author(s): Julia Mckenna, Fabiana Bisaro, Addison Lueck, Andrea Russell, Dean Crick
Presenter(s): Julia Mckenna
Mentors(s): Miriam Braunstein
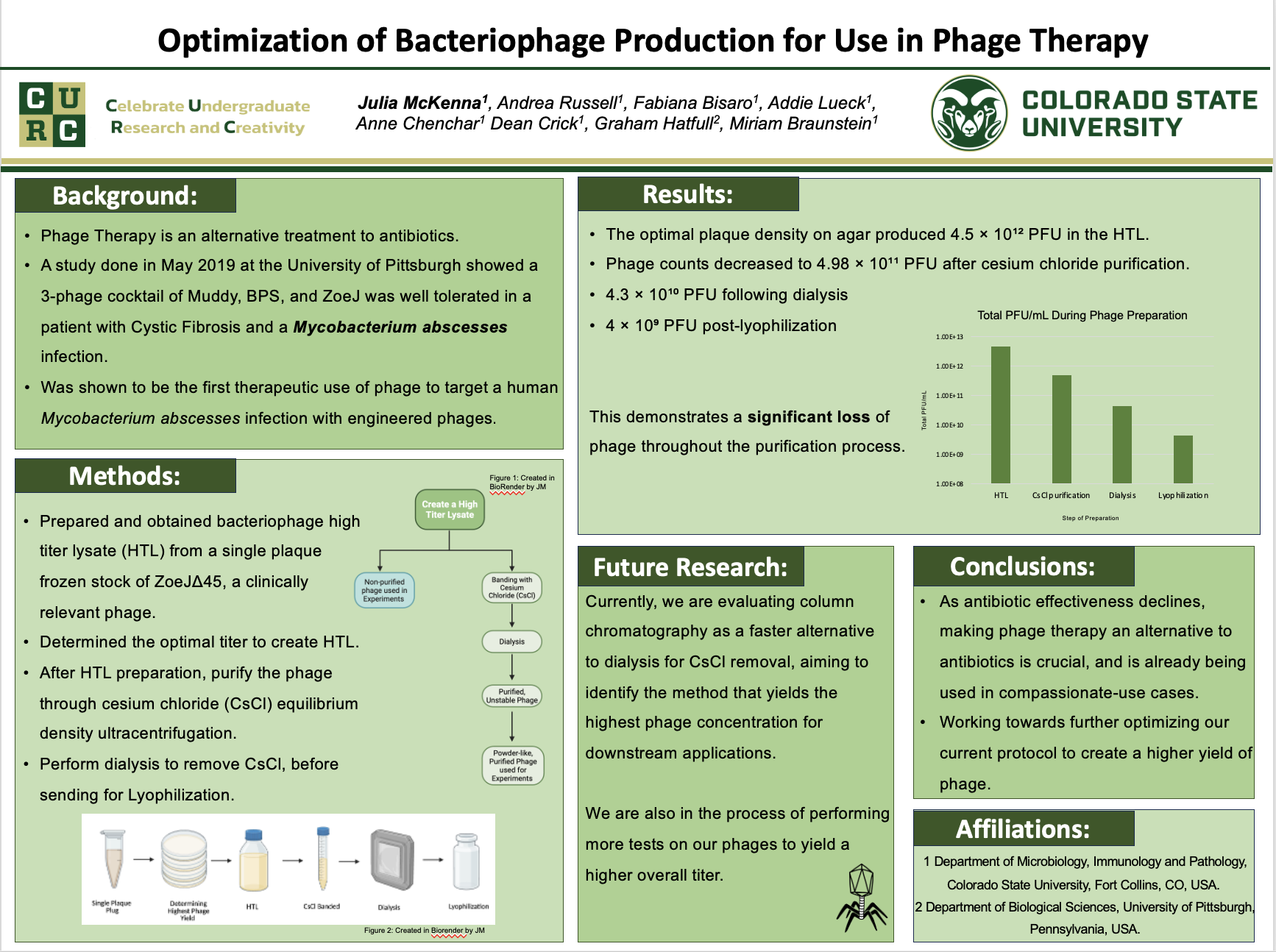
Multi-drug resistant Mycobacterium abscessus is a nontuberculous mycobacterium (NTM) that represents a global threat to patients with pulmonary disease. Bacteriophages are viruses that infect and kill bacteria. Therapeutic use of bacteriophages (i.e., Phage therapy) offers a promising alternative to antibiotics and is being used in compassionate use cases. To prepare phages, we do the following: 1) amplify them in bacteria on agar plates to produce a high-titer lysate (HTL), 2) purify the phage through cesium chloride (CsCl) equilibrium density ultracentrifugation, 3) remove CsCl by dialysis, and 4) lyophilize the phages. Here, we aim to optimize the protocol, ensuring that our phage preparation is pure and at a high concentration suitable for in-vitro and in vivo phage therapy studies. We prepared HTL of ZoeJΔ45, a clinically relevant phage, from a single phage plaque. Next, we optimized/identified the number of plaques on an agar plate that produced the highest yield of total phage in the HTL, which was 4.5×10^12 plaque formation units (PFU) of phage. After CsCl purification, the total number of phages decreased to 4.98×10^11 PFU, and it dropped further to 4.3x1010 PFU after dialysis. After lyophilization, there were 4x10^9 PFU remaining. The results show loss of phage throughout the purification process, We are evaluating column chromatography as an alternative to remove CsCl more quickly than dialysis, potentially increasing phage yield. Our goal is to compare purification strategies to identify the method that produces the highest phage yield for downstream applications.
